The atomic theory4/18/2024 This allowed calculation of its mass as tiny, equal to 9.109 * 10 −31 kg. 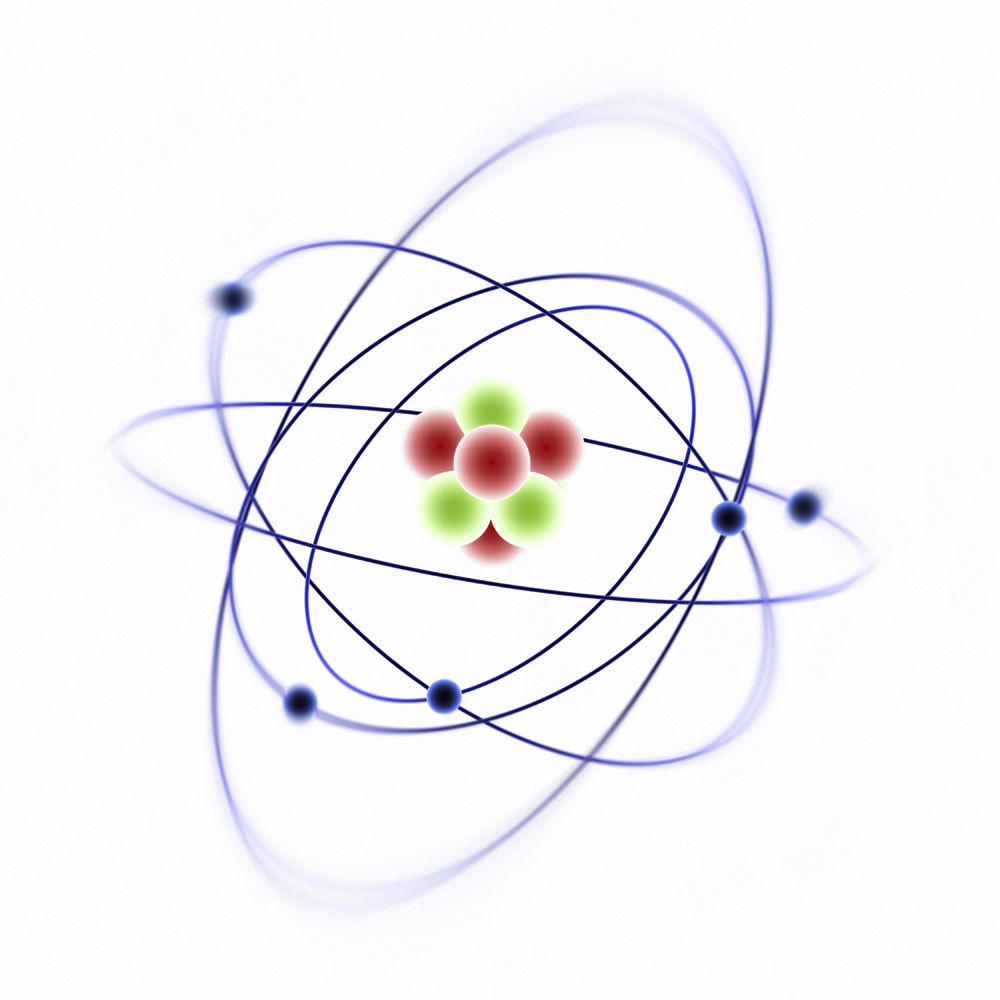
In 1906, Robert Millikan determined that the electrons had a Coulomb (C) charge of -1.6 * 10 −19. It was described as a positively-charged sphere in which the electrons were inlaid (with negative charges). The negative charges were named electrons ( e-).Īccording to the assumptions established about the atom's neutral charge, Thomson proposed the first atomic model. He established that, for this to occur, an atom should have the same quantity of negative and positive charges. Thomson knew that the atoms were electrically neutral. Crookes named the emission ' cathode rays'.Īfter the cathode ray experiments, Sir Joseph John Thomson established that the emitted ray was formed by negative charges, because they were attracted by the positive pole. When creating a vacuum in the tube, a light discharge can be seen that goes from the cathode (negatively-charged electrode) to the anode (positively-charged electrode). In 1850, Sir William Crookes constructed a ' discharge tube', that is a glass tube with the air removed and metallic electrodes at its ends, connected to a high- voltage source. Thomson's atomic model Schematic representation of the Thomson model. Together, these were good evidence that atoms existed.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
:max_bytes(150000):strip_icc()/GettyImages-1157225833-01064c770b904b23bb07df6eec68f35d.jpg)

 RSS Feed
RSS Feed
